FiberLocker® System
Mechanical Augmentation of Rotator Cuff Repair through Fiberlocking
To fiberlock™
fi·ber·lock | \ˈfaɪbɚˌlɑk\ verb
To reinforce soft tissue, typically tendon, by interweaving patch fibers into tissue, resulting in immediate mechanical strength to the repair construct.
The FiberLocker® System is commercially available in the U.S.
FiberLocker® Instrument

SpeedPatch®

Ease of Use
The simple implantation is completed in just 90 seconds.1
Immediate Mechanical Strength
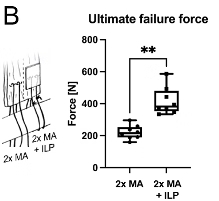
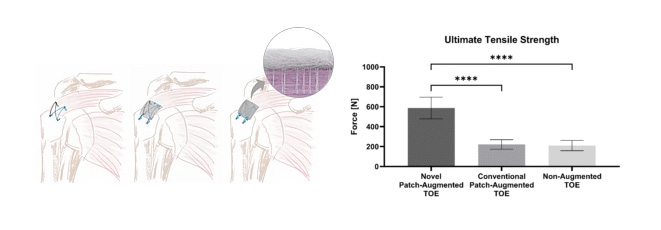
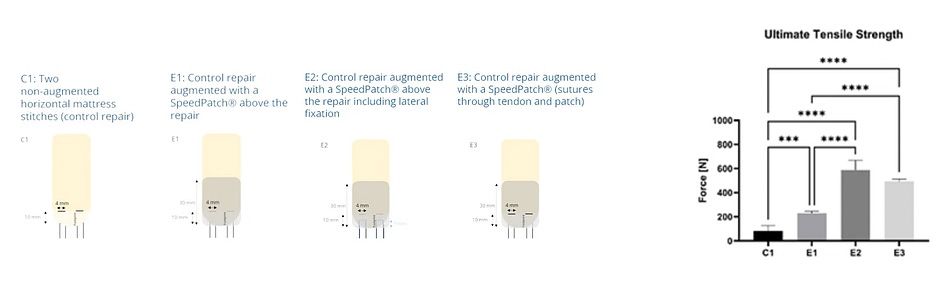
Ex vivo testing has shown a significantly increased biomechanical strength of augmented rotator cuff repairs at time zero.2,3
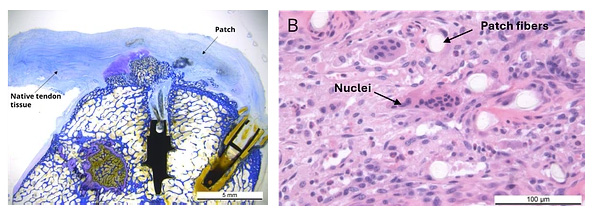
Cellular Ingrowth
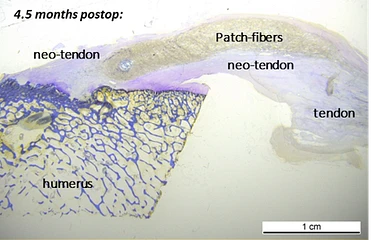
Animal studies demonstrated cell infiltration into the porous scaffold.2,4
Clinical Application
Clinical Challenge
Rotator cuff tears affect between 20.4% and 34% of the general population.5-7 Despite surgical repair, retear rates remain high—ranging from 24% to 94%.8,9 Suture cut-through, where sutures pull through the tendon tissue, is the primary cause of failure, accounting for 86% of all cases.10 This highlights the compromised mechanical integrity of the repaired tendon. Until now, no augmentation system has provided sufficient mechanical reinforcement to limit tendon retears and support healing effectively.
Technology
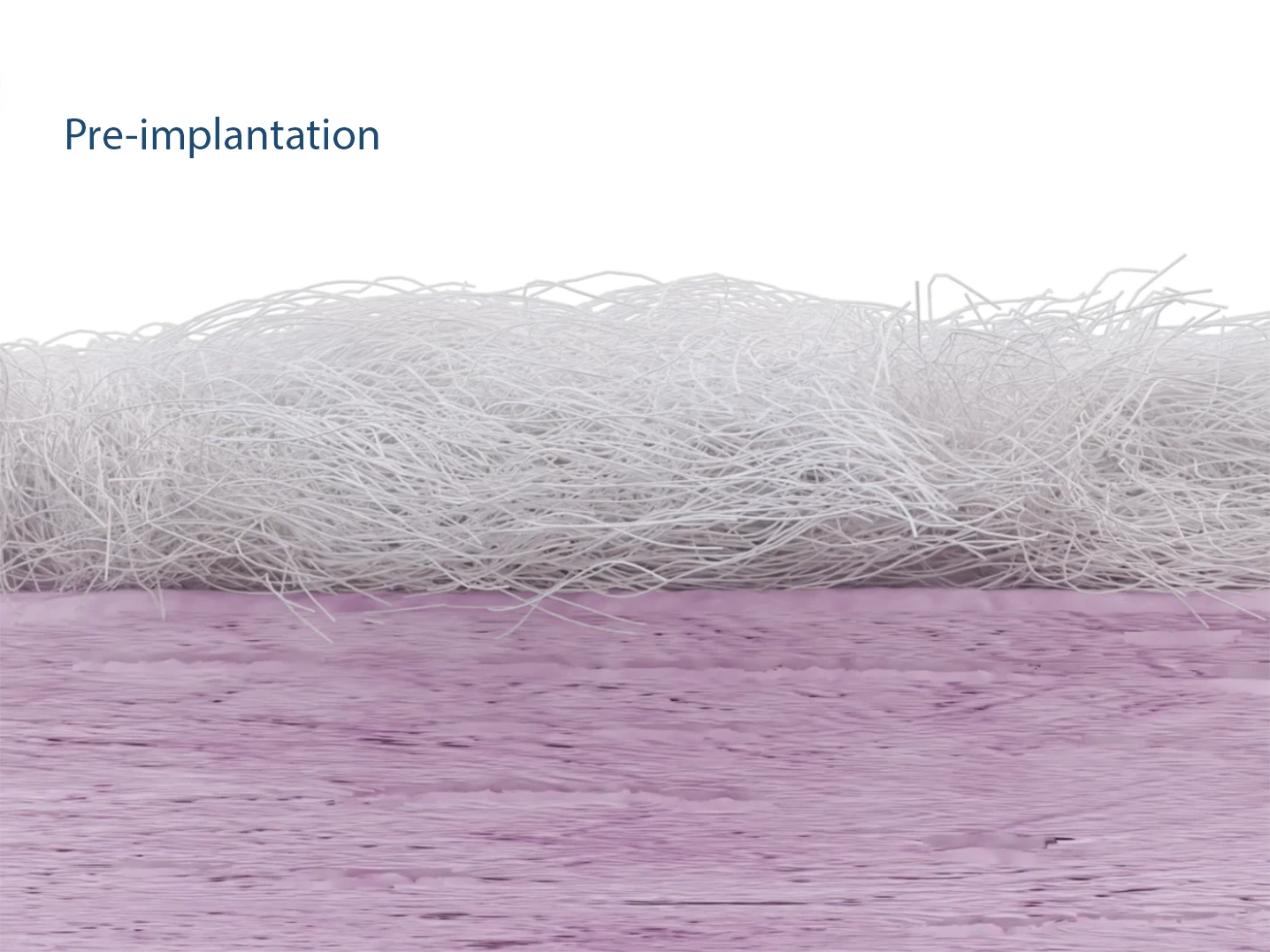
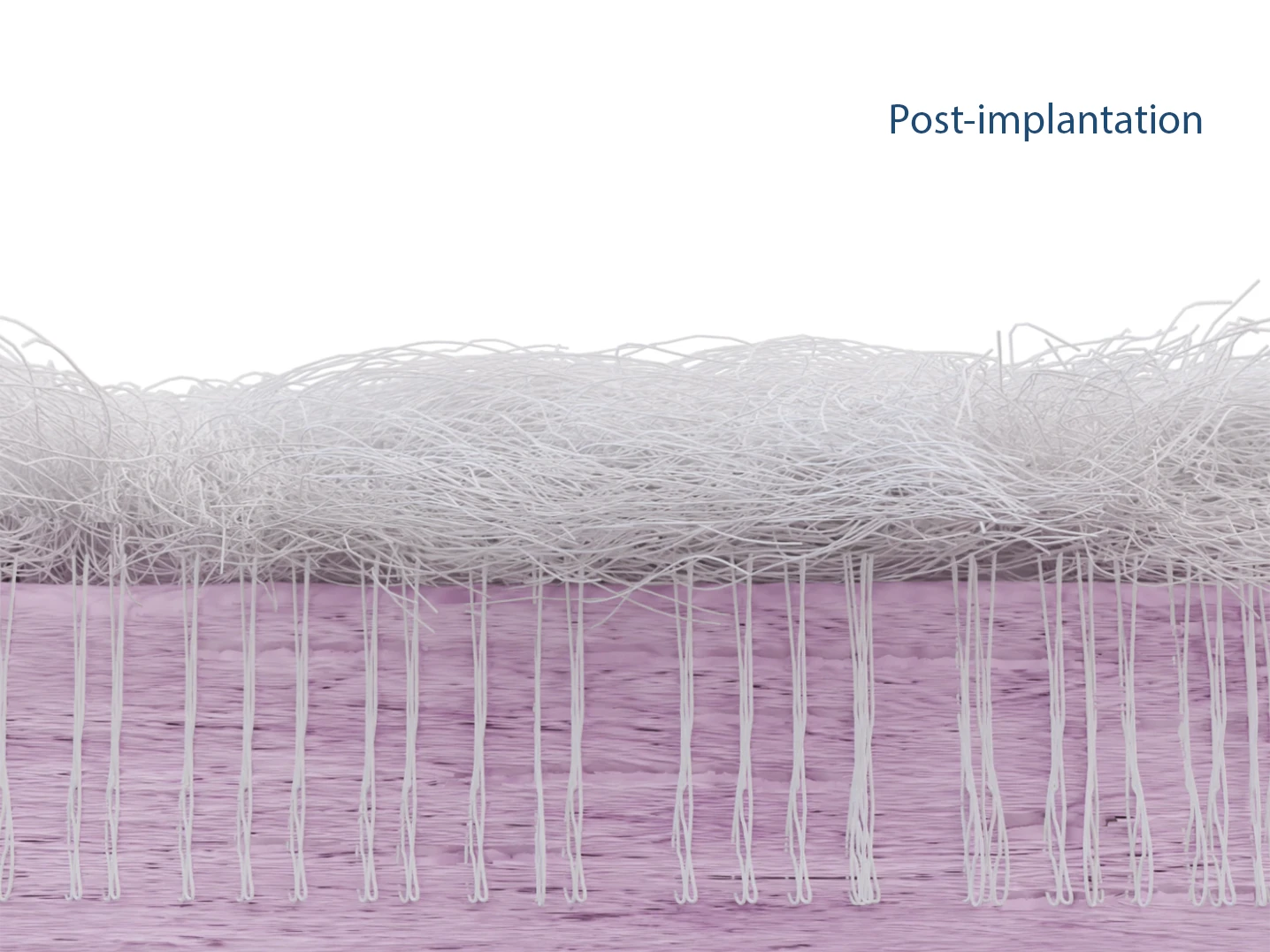
The patented Fiberlock™ technology enables the formation of a strong, interwoven network between the patch and tendon tissue. During implantation, the instrument’s reciprocating needle pushes implant fibers into the underlying soft tissue, securing the patch across the entire patch–tendon interface. This results in immediate mechanical augmentation.2,3
Surgical Procedure
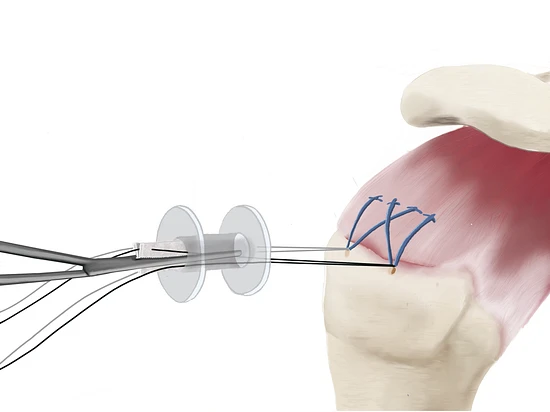
1. Delivery
During or after the repair of the rotator cuff tendon, the SpeedPatch® can be easily delivered according to each surgeon’s preference.
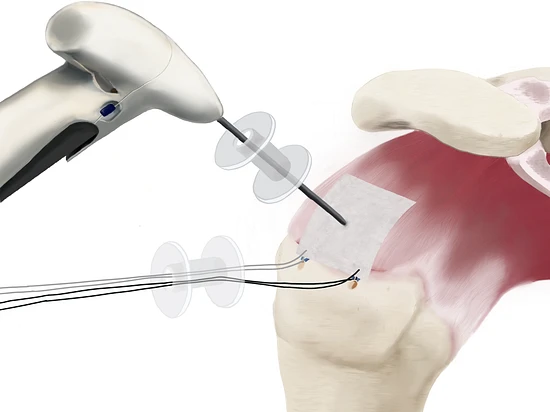
2. Implantation
The SpeedPatch® is secured to the tendon tissue using the FiberLocker® Instrument. In just 90 seconds, the felting technology enables a secure implantation across the entire patch-tendon interface.

3. Lateral Attachment
Lateral patch-to-bone fixation using sutures and anchors completes the construct.
Scientific Evidence
Biomechanical Data
Biological Data
Clinical Data
Currently, two prospective clinical studies investigating the performance and safety of the FiberLocker® System are ongoing.
First-in-Human Study Switzerland
Conducted at Balgrist University Hospital, this study marks the first clinical use in Switzerland, investigating the safety of the FiberLocker® System.
Multicenter Study in the U.S.
The primary outcome of this study is healing based on MRI, and it is being conducted across multiple centers in the United States.